|
It is now well-established that this effect is underpinned by a unique glucose transporter, GLUT4, expressed in insulin-sensitive cells. The insulin-dependent increase in glucose transport in peripheral tissues (muscle, adipose) is a key action of insulin 1, 2. Here we show that both GLUT4 delivery to the plasma membrane and GLUT4 dispersal are reduced in larger adipocytes, supporting the hypothesis that larger adipocytes are refractory to insulin challenge compared to their smaller counterparts, even within a supposedly homogeneous population of cells. Adipocytes of different sizes have been shown to exhibit distinct metabolic properties: larger adipocytes exhibit reduced insulin-stimulated glucose transport compared to smaller cells. Consistent with dispersal being an important physiological mechanism, we observed that insulin-stimulated GLUT4 dispersal is reduced under conditions of insulin resistance. 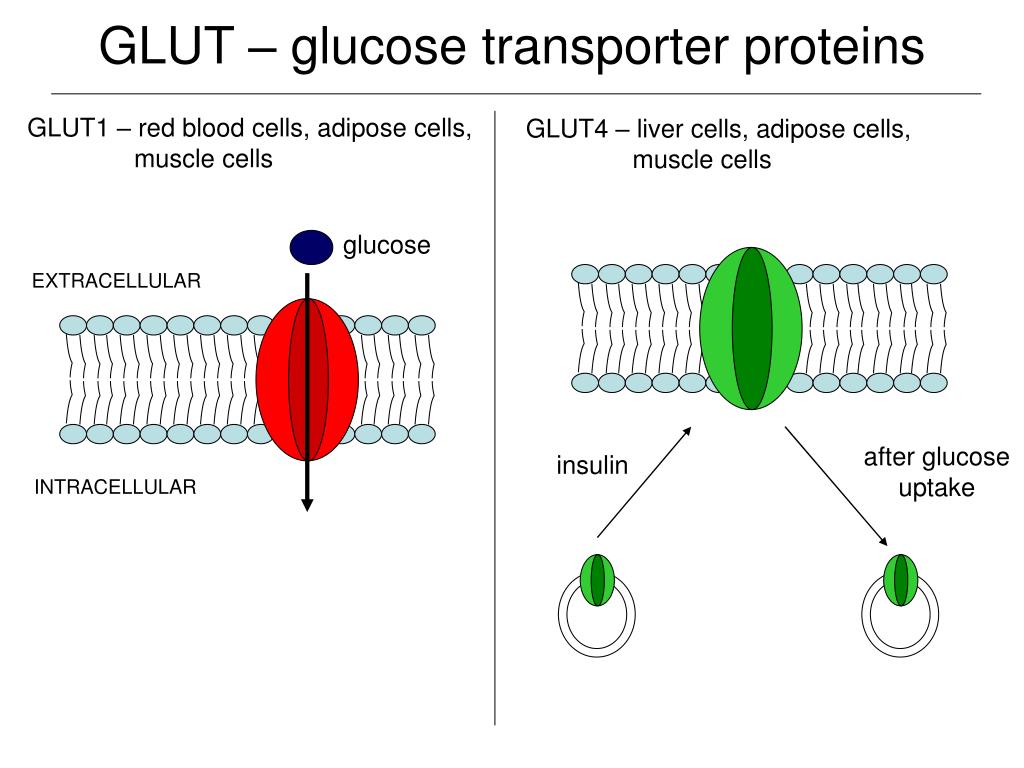
By contrast, insulin-stimulated dispersal of TfR was not observed in HeLa cells, suggesting that the mechanism may be unique to GLUT4. Here we show that insulin stimulates GLUT4 dispersal in the plasma membrane of adipocytes, induced pluripotent stem cell-derived cardiomyocytes and HeLa cells, suggesting that this phenomenon is specific to GLUT4 expressed in all cell types. Although shown in adipocytes, whether insulin-stimulated dispersal occurs in other cells and/or is exhibited by other proteins remains a matter of debate. Recent studies have revealed that a further action of insulin is to mediate the dispersal of GLUT4 molecules away from the site of GLUT4 vesicle fusion with the plasma membrane. This is underpinned by the delivery and fusion of GLUT4-containing vesicles with the plasma membrane. 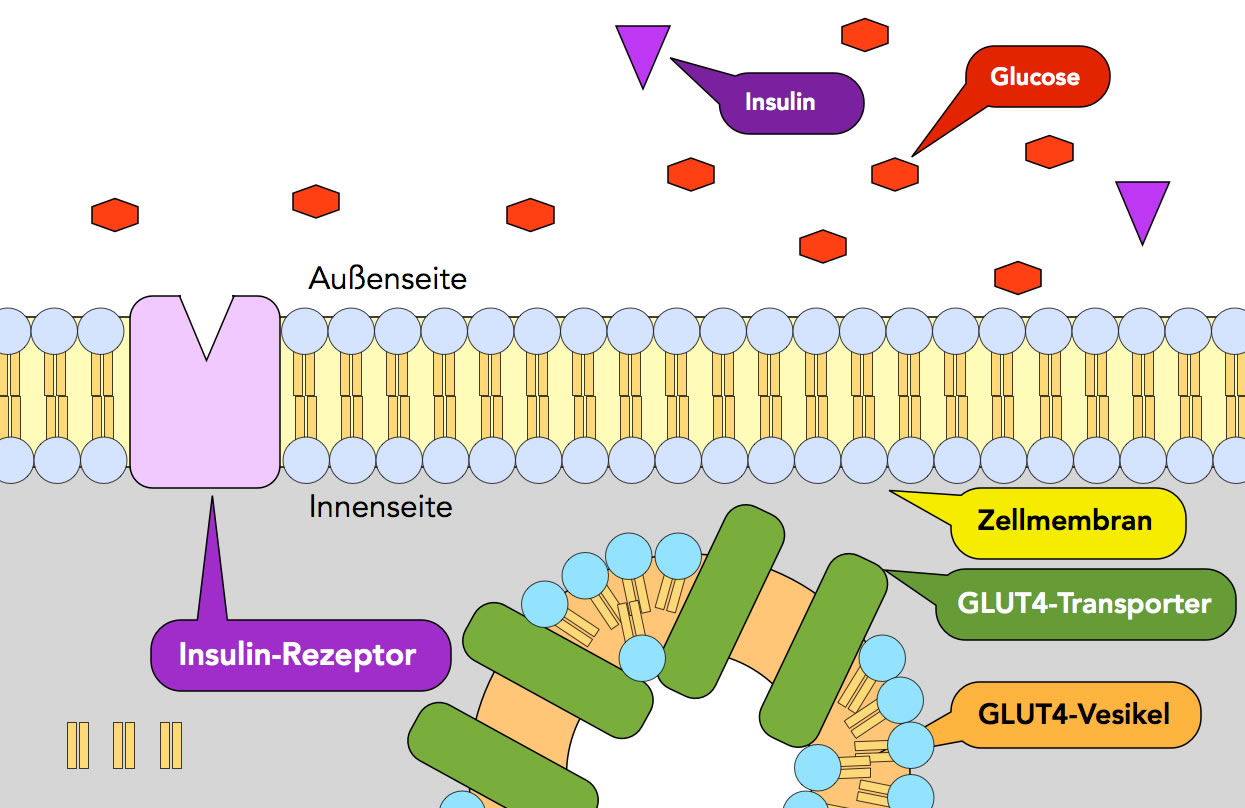
The regulated translocation of the glucose transporter, GLUT4, to the surface of adipocytes and muscle is a key action of insulin.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
